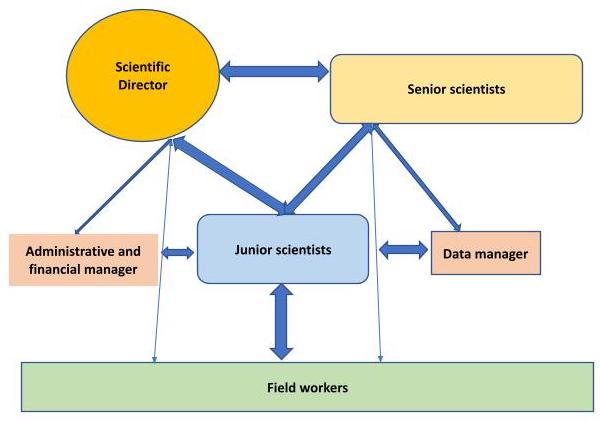
Data Management

Led by Prof Issaka Sagara
This unit ensures :
- Database design
- Vaccine trials
- Drug Trials
- Investigations
- Database management
- Collaboration with other institutions (Muso, CICM, UCRS, IRSS Bobo, Manferinya, Guinea) for data management.
- Data management training (workshops and courses)
Management software and tools:
- Stata, SPSS, R etc
- Platforms (data collection via tablets)
- Share point
- Internet Connexion (Afribone) since 2015
- Data server (mrtc-data)
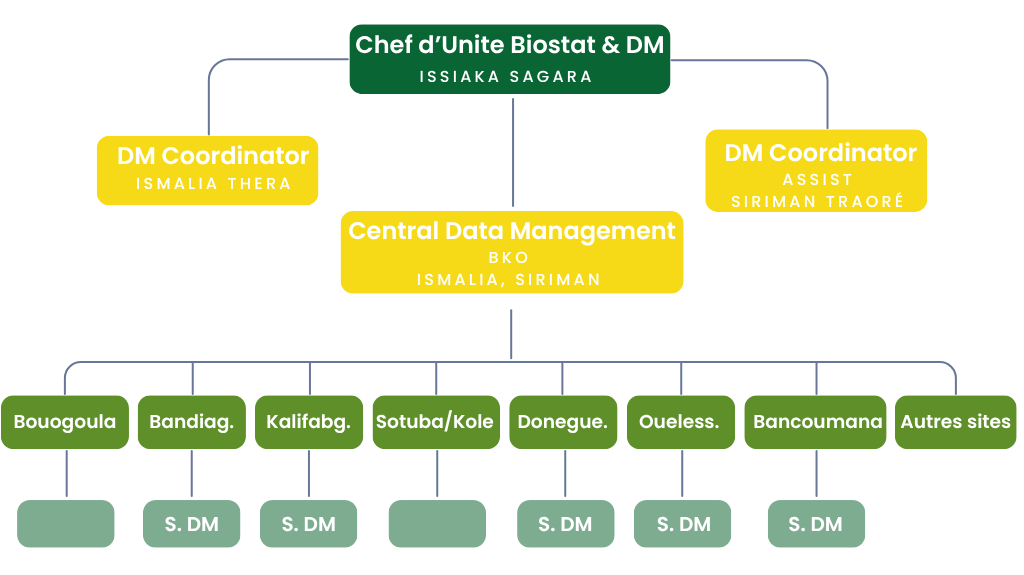
Data Management Organization Chart

Led by Pr. Doumbo Safiatou Niaré
Initiated in 2012, this unit was created in 2019 through collaboration with: “Hôpital Dermatologique de Bamako » (Ex CNAM), ESTHER (microsporidiose), France. It ensures :
- Satisfying the needs and requirements of patients and health professionals, and to provide them with a quality of service equal to that of fungal disease diagnosis.
- Guarantying and continuously improve the reliability of mycological test results.
- Developing the competence and expertise of the laboratory staff in the field of mycological diagnosis.
- Improving the operating efficiency of the mycology laboratory.
- Building collaboration with health facilities and laboratories: support for cares.
- Continuing education in mycology: Thesis, presentations and theoretical and practical training in mycology.
- Writing and regular execution of fungi Research Grants.
The technical tray includes: fungal culture chain and identification, an incubator, a host, 2 microscopes, a bench, laboratory materials and reagents essential for direct examination and fungi growth.
The laboratory has just acquired a Mass spectrometer VITEK MS (Maldi-Tof), which will allow it to reinforce the diagnostic techniques and to be more specific and sensitive in the results of biological materials.
To ensure the safety and security of results Our laboratory has specially adapted facilities, which guarantee an optimal analysis of all specimens.
The laboratory staff is committed to doing everything possible to ensure the protection, security and integrity of data for reliable results.
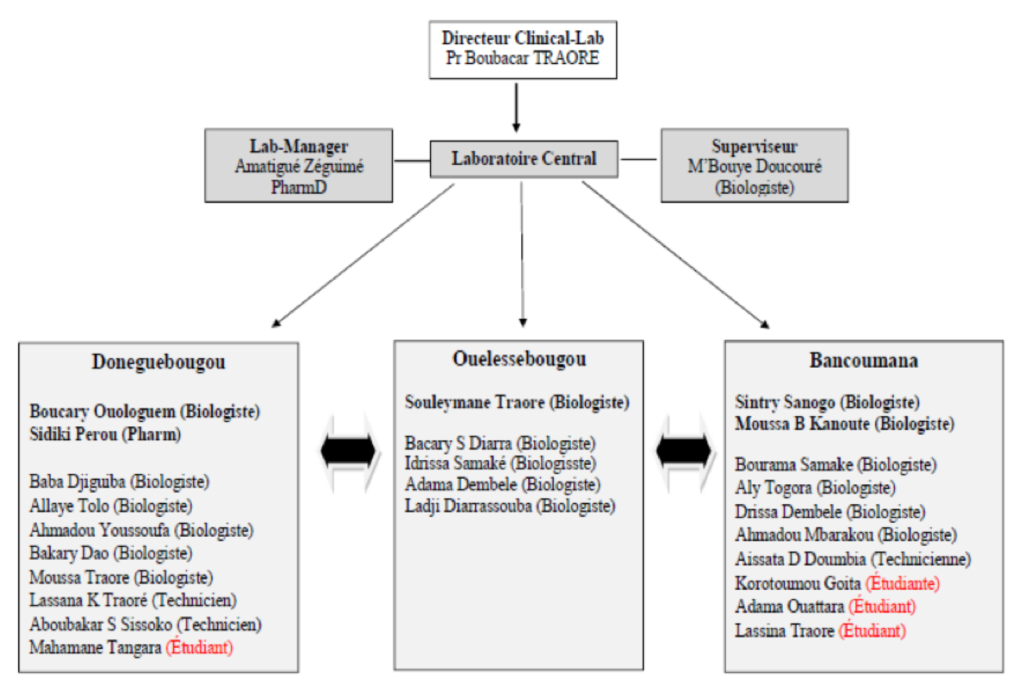
Dirigé par Pr. Boubacar Traoré / Dr Amatigué Ziguimé
Created in 2001, the Clinical Laboratory establishes frameworks for Good Laboratory Practice (GLP) and CLIA in Clinical Biology (Establish GLP and CLIA in clinical biology areas):
- Hematology
- Biochemistry
- Parasitology
- Serology / Immunology
The Clinical Laboratory supports clinical trials of vaccines and other clinical trials to assess the eligibility of volunteers, vaccine tolerance, efficacy, immunogenicity of vaccines. The clinical laboratory was accredited in April 2010, October 2014, October 2016, October 2018. The technical tray is made of :
- A hematology machine (DxH500) and biochemistry machine (Pentra C 200) at each site (maintenance/ calibrations at least twice a year)
- Cold chain (cold room, freezers …)
- Sample/Reagent Management System (Freezer Work) and “Vaisala for Temp monitoring”)
- Machines for ELISA and other equipment (Maintenance/ calibration, correlation)
- Direct data entry (iDataFax), tracking of abnormal values and other
- In perspective: qPRC for soon.
Clinical Lab Organization Chart
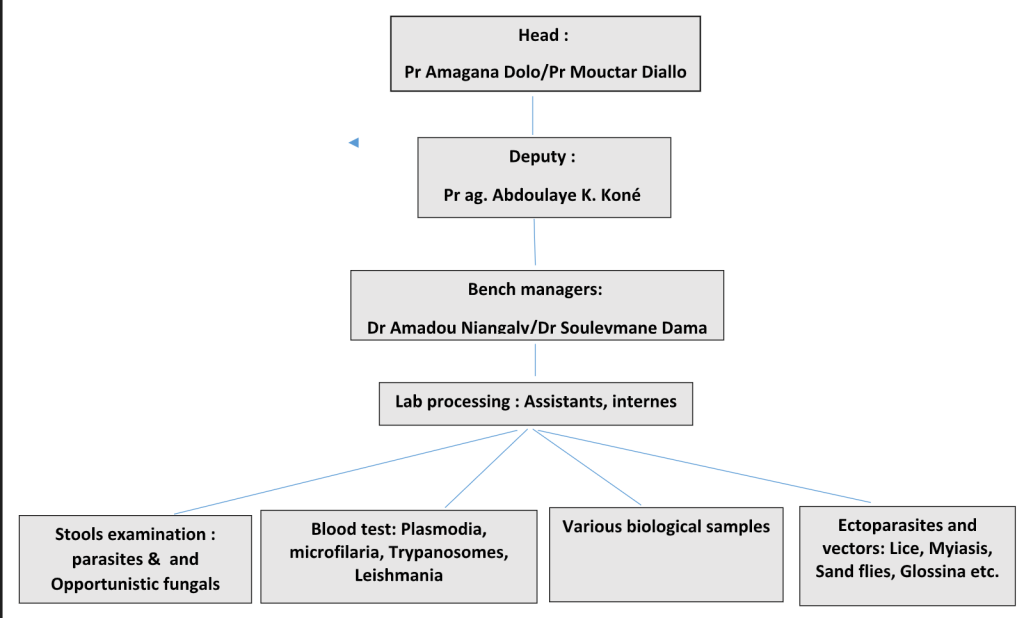
Parasitic diagnostic unit
Led by Prof Amagana Dolo / Prof Mouctar Diallo / Pr Agrégé Abdoulaye Kassoum Koné
Created at the same time as the Department of Epidemiology of Parasitic Diseases (DEAP); it is the oldest unit of the MRTC.
It was set up to carry out the following missions :
- Ensure quality control of diagnosis of parasitic diseases (care services, support to control programs, sentinel sites – PMI)
- Introduce interns, master degree candidates (medicine, pharmacy, biology) to basic parasitology techniques
- Train staff (technician, physician, etc.) in parasitic diagnostic techniques and identification
- Support to research by processing and delivering results for specific research purposes,
- Test and validate the parasitic diagnostic techniques
The activities are:
- Diagnosis of sanguicoles parasites,
- Diagnosis of stool parasites,
- Diagnosis of ectoparasites
- Diagnosis of opportunists (stool parasites and fungus)
- Training and Capacity building: collection and sharing of parasites specimens for laboratory practices.
Chart
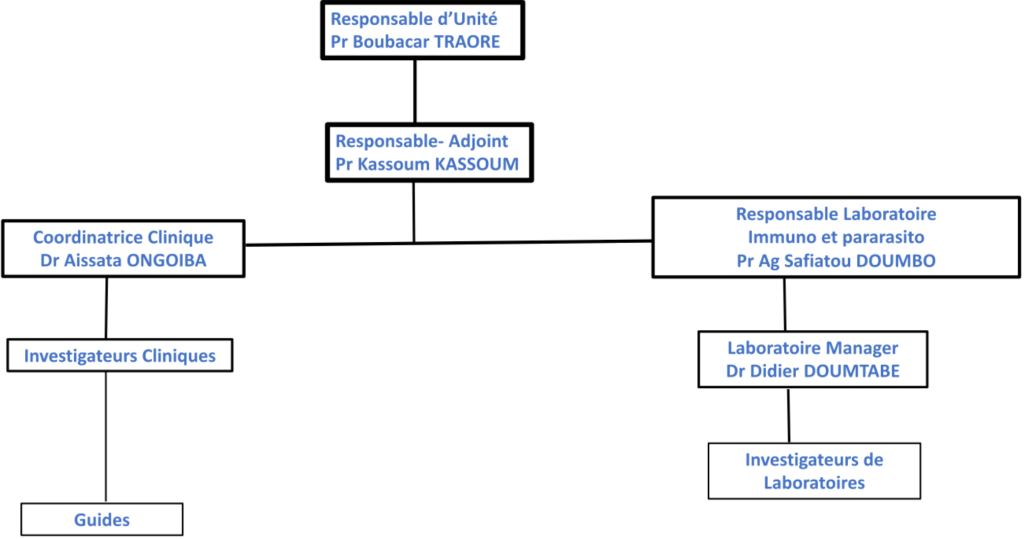
PREMA
Led by Prof Boubacar Traoré / Prof Kassoum Kayentao
This unit was formally created in 2006 from the team that worked on the project « IPTp-SP ou CQ versus CQ weekly» in 1998-2001 among pregnant women reinforced by the return of Prof Boubacar Traoré in 2003.
Mission
- Contribution to the evaluation and implementation of malaria prevention strategies during pregnancy,
- Contribution to the understanding of the pathophysiology of malaria: immunology of placental infection,
- Contribution to the understanding of immune response acquisition during natural infestation with P. Falciparum,
- Promotion of training in the fields of parasitology, parasitic immunology
PREMA Organization Chart
Immunology
Led by Prof Bourèma Kouriba
PACEMI works on the following research topics :
- Immunology of parasitic diseases;
- Immunology of emerging and re-emerging viral pathologies;
- Genetics of parasitic diseases;
The technical tray includes: fungal culture chain and identification, an incubator, a host, 2 microscopes, a bench, laboratory materials and reagents essential for direct examination and fungi growth.
The laboratory has just acquired a Mass spectrometer VITEK MS (Maldi-Tof), which will allow it to reinforce the diagnostic techniques and to be more specific and sensitive in the results of biological materials.
To ensure the safety and security of results Our laboratory has specially adapted facilities, which guarantee an optimal analysis of all specimens.
The laboratory staff is committed to doing everything possible to ensure the protection, security and integrity of data for reliable results.
Staff
- Pr Bourèma Kouriba PharmD, MSc, PhD, Associate Lecturer of CAMES, Head of Unit, Teacher-Researcher, USTTB, Ministry of Health
- Dr Charles Arama PharmD, MSc, PhD, Research Officer, CAMES Master Assistant, USTTB, Ministry of Health
- Dr Issa Diarra PharmD, MSc, Research Associate, USTTB, Ministry of Health
- Dr Karamoko Niaré PharmD, MSc, PhD Candidate, in training at KEMRI, Kenya and WACCIP, Ghana
- Dr Amatigue Zeguimé PharmD
- M. S. Yirampo MSc, Investigator
- Dr K. Bangaly PharmD, MSc candidate, investigator and vaccination team
- Dr H. Doucouré PharmD, Investigator
- M. B. Koné PharmD candidate
- Me. O. Sissoko PharmD candidate
Schistosomiasis

Led by Pr. Abdoulaye Dabo
The DEAP has been a WHO Collaborating Centre for Schistosomiasis since 1987. The creation of UFR Schistosomoses and Molluscs Intermediate Hosts was created in 1995. The unit has as mission:
- Generate research results on schistosomosis, helminthiasis and intermediate host molluscs for use in informed public health decision-making;
- Test efficient and generalizable control strategies for schistosomosis, helminthiasis and intermediate host molluscs;
- Enhance the training (interns, trainees, assistants, PhD students) and research (Masters and PhD) potential of the learners receiving MRTC/DEAP/FMPOS.
Vaccine

Led by Prof Issaga Sagara
MEDRU

Led by Pr. Abdoulaye DJIMDE
Laboratories and their role within the parent organization
The MEDRU laboratories are part of the MRTC/DEAP and have been in operation since 2001.
A thorough understanding of our laboratory’s objectives for our employees is essential. We continuously train, update and engage employees based on our mission and the services we provide to our partners. We assess the capabilities and expertise of our human resources (HR) and employees in light of our missions and the objectives and requirements of our partners.
At each specific activity, appropriate persons are designated to carry out this task. Designated individuals will manage reagent, consumable, inventory and order requirements. These individuals are responsible for sample processing, collection, data analysis and the development of the study report. Data management includes protocol approval, record keeping and electronic media to ensure data accuracy and avoid bias. The data is saved in the PCR laboratory and also saved on the DELGEME server by our IT manager. Access to data is limited to employees and partners if necessary. Access to samples and data is restricted to authorized personnel (high confidentiality, password protection, lock-in of archives, restriction of access to data).
Access to samples and data is restricted to authorized personnel (high confidentiality, password protection, lock-in of archives, restriction of access to data). The MEDRU/MRTC/DEAP Laboratory Manager works with the designated individuals to regularly evaluate our operation and take corrective action to ensure good performance at each step of the procedures.
- The missions of our laboratories are :
- Basic and applied research (The main laboratory MEDRU/MRTC/DEAP
- Point-G and the laboratory of Sotuba)
- Drug discovery
- Drug resistance
- Cell Biology
- Genomics and Bioinformatics
- Pharmacology [PD, PK, PG]
- Nutrition
- Entomology
- Clinical research (MEDRU/MRTC/DEAP-Sotuba, Bougoula, Kolle, Djoliba, Faladie)
- Clinical Trials
- Field observation studies
- Design of studies
- Internal Monitoring
- Partner and Client Services (MEDRU/MRTC/DEAP-Point-G and Sotuba Laboratory)
- Sample Management
- Analysis of Samples
- Report writing
- Data Management and Sharing (The MEDRU/MRTC/DEAP-Point-G main laboratory and the Sotuba laboratory)
- Scientific Commission (All MEDRU/MRTC/DEAP laboratories)
- Search for funding opportunities
- Scientific Writing and Valorisation of Research
- Training
- Basic and applied research (The main laboratory MEDRU/MRTC/DEAP
- Laboratory Activities and Services
To achieve its objectives, MEDRU laboratories have the following platforms:- Clinical Trials: (The MEDRU/MRTC/DEAP- sites of Bougoula, Djoliba, Faladie, Kolle, Sotuba) Classical Polymerase Chain Reaction (PCR) and quantitative PCR for molecular diagnosis (The main laboratory MEDRU/MRTC/DEAP-Point-G and the laboratory of Sotuba)
- Advanced biochemistry and hematology machines are used to measure the following parameters: ALAT, ASAT, Glucose, Total and Direct bilirubins, Creatinine, ions (potassium, chlorine, calcium, magnesium), blood count (The MEDRU/MRTC/DEAP laboratories in Bougoula, Kolle and Sotuba)
- Cell cultures are conducted to understand the in vitro/ex vivo resistance mechanism and for the screening of new drugs (The main laboratory MEDRU/MRTC/DEAP-Point-G). Insectarium (Captures of vectors and experimental infestations are carried out to evaluate the parameters related to transmission in collaboration with our entomologist colleagues). (The main laboratory MEDRU/MRTC/DEAP- Point-G and the laboratory of Bougoula)
- High Performance Liquid Chromatography (HPLC) machines are used for the dosing of drugs and their metabolites (The main laboratory MEDRU/MRTC/DEAP- Point-G). Impedance meter and FTIR for nutritional assessment (The main laboratory MEDRU/MRTC/DEAP- Point-G)
- A platform for high-throughput sequencing (The main laboratory MEDRU/MRTC/DEAP- Point-G)
Organizational chart

Ouelessebougou Clinical Research Unit
Led by Prof Alassane Dicko
Brief history :
- Epidemiology Biostatistics and Data Management Unit (2001 –
- Site Preparation for Malaria Vaccine Testing (1998-2003)
- MVDB Malaria Vaccine Test (former LMIV): Doneguebougou, Bancoumana
- 2007: Strategic withdrawal of vaccine trials and NIAID programmes for the development of a new site in Ouelessebougou
- 2008: Ouelessebougou Clinical Research Unit.
Malaria Vaccine Clinical Development Unit (MVCDU) / Bandiagara Malaria Project (BMP)

Led by Prof Mahamadou A Thera
The Bandiagara Malaria Project was initiated as an NIH-supported collaboration between the Malaria Research and Training Center (MRTC) and the University of Maryland’s Center for Vaccine Development and was launched in 1998. The initial main aim was to develop sites and prepare them to conduct malaria vaccine trials under international standards. BMP research scope covered all aspects of malaria research with focus on clinical trials of malaria vaccines. To achieve this a better understanding of malaria epidemiology and characteristics of population at risks were necessary. Hence, BMP established normal reference ranges of Malian adult population in Bandiagara and investigated the clinical epidemiology of malaria infection and diseases.
Later, the research program enlarged its scope to include host-pathogens interactions, severe malaria pathogenesis, gut microbiome influence, genomic epidemiology of malaria, impact of large-scale malaria control strategies. In addition, a large focus was given to emergent viral fevers.
BMP has been acknowledged in 2011-2016 by the African Network for Drugs and Diagnostics Innovation (ANDI) as an ANDI Center of Excellence for Clinical Development of Malaria Products.
BMP missions are :
- To characterize malaria epidemiology and genomic epidemiology
- Investigate malaria parasites and human host interactions to understand disease pathogenesis
- Contribute to the Clinical Development of drugs and vaccines by undertaking ICH/GCPs compliant clinical trials
- Reinforce local health care system capacity to address local health problems
- Guide and inform Mali National Malaria Control Program’s control strategies implementation by maintaining a sentinel site for malaria drug efficacy surveillance and others malaria studies
- Help ensure integrity and research productivity of the MRTC
- Contribute to the surveillance and understanding of emerging pathogens
Under the leadership of Pr Mahamadou A. Thera, BMP has developed to become a whole research structure with a well-established research center in the field in Bandiagara and a Clinical Trial Center in Bamako, Point G.
Prof Thera is seconded by Dr Drissa Coulibaly, Pr Abdoulaye K Kone and Pr Karim Traore, the senior researchers, a team of twelve junior scientists and fourteen support personnel.
Leadership

Mahamadou A Thera
Biosketch
Mahamadou A Thera, MD, MPH, PhD, FAAS, is the Scientific Director of the Bandiagara Malaria Program (BMP) at the Malaria Research and Training (MRTC) of the University of Sciences, Techniques and Technologies of Bamako, in Mali. Since 2008, he is Professor of Parasitology-Mycology at the Faculty of Medicine and Odonto-Stomatology of Bamako.
He was principal investigator of several clinical trials that assessed blood-stage malaria vaccine candidates and malaria drugs. His group established for the first time, proof of concept of the allele specific protective efficacy of a blood-stage malaria vaccine against clinical malaria in a malaria endemic country. Pr Thera also directed several epidemiological studies of malaria in Mali.
He has been member of several scientific advisory committees including the WHO advisory body for malaria vaccines MALVAC and JTEG, the European Vaccine Initiative Scientific Advisory Committee (EVI-SAC), that he chaired during the period 2015-2017. He has co-authored more than 110 manuscripts indexed in PubMed.
Pr Thera holds a MD from the Institute of Medicine and Pharmacy of Jassy, (Romania, 1986), a MPH from University of Bamako (Mali, 1996), a MSc in Biostatistics and Conduct of Clinical Trials from University of Paris 6 (France, 2003) and a PhD in Parasitology-Mycology from University of Bamako’s Institute of Training and Applied Research (ISFRA) (Mali, 2015).
Dr Thera was in 2000, the first Fellow of the WHO/TDR Clinical Research and Development Fellowship, initially supported by Glaxo SmithKline Biologicals and TDR. This fellowship turned into a successful capacity building program in Clinical research and product development for developing countries, thanks to substantial funding received from the Gates Foundation and EDCTP.
As Professor at the FMOS, Pr Thera contribute to train undergraduate MDs, and PharmDs, post graduate MSc and PhD students, in Mali and in the neighboring countries.
Pr Thera is member of the Académie des Sciences du Mali (ASM) since 2018 and a Fellow of the African Academy of Sciences since 2020.
Dr Thera’s areas of expertise include malaria epidemiology, malaria pathogenesis, clinical trials, product clinical research and development, ethics, parasitic diseases.
Actual work
Has two main directions. First direction addresses issues related to human host-malaria parasites interactions. It focuses on understanding the role of variant PfEMP1s antigens in the pathogenesis of severe malaria, either cerebral malaria or severe malaria anemia. This is done by analyzing seroreactivity profiles to microarrays of antigens printed on slides, of children aged under five years in a case-control study of severe malaria. This project was supported by the NIH R01AI099628, awarded to Prof Thera in 2014-2020. The second direction of work is focused on the clinical development vaccines against malaria and viral infections. The malaria vaccine candidate derived from P. falciparum merozoite surface protein 3, the MSP3-CRM candidate manufactured by Vac-4-All is being tested in a series of clinical trials to demonstrate the proof of concept of its safety, immunogenicity and efficacy against clinical malaria.
Selected publications
(List of Published Work in MyBibliography: https://www.ncbi.nlm.nih.gov/myncbi/1VMcnFoclLkks/bibliography/public/)
- Thera MA, Kone AK, Tangara B, Diarra E, et al., School-aged children based seasonal malaria chemoprevention using artesunate-amodiaquine in Mali. Parasite Epidemiology and Control. 2018. doi.org/10.1016/j.parepi.2018.02.001
- Djimde A, Ranque S, Thera MA. Building a center of excellence in biomedical research in an unfavorable environment: the Malaria Research and Training Center in Mali. Med Sante Trop. 2019. doi: 10.1684/mst.2019.0938. PubMed PMID: 31884979.
- Malaria Genomic Epidemiology Network, Band G, Rockett KA, Spencer CC, Kwiatkowski DP. A novel locus of resistance to severe malaria in a region of ancient balancing selection. Nature. 2015. doi:10.1038/nature15390.
- Thera MA, Doumbo OK, Coulibaly D, Plowe CV et al., A field trial to assess a blood-stage malaria vaccine. N Engl J Med. 2011. doi: 10.1056/NEJMoa1008115.
- Tishkoff SA, Reed FA, Friedlander FR, Ehret C, Thera MA, Wambebe C, Weber JL, Williams SM et al., The genetic structure and history of Africans and African Americans. 2009. doi:10.1126/science.1172257.

Dr Drissa Coulibaly
Biosketch
Drissa Coulibaly is a physician and specialist in parasitology with extensive experience in pediatric studies in Mali, particularly Bandiagara. He conducted multiple malaria epidemiology studies in over the past two decades, including cross-sectional surveys, case-control studies, and longitudinal cohort studies. He was involved in developing Bandiagara as a malaria vaccine testing site in Mali since 1998 and has led multiple NIAID-sponsored studies there. He has expertise in the implementation of malaria vaccine clinical trials according to international standards. He successfully conducted Phase 1 and Phase 2 clinical trials using candidates malaria vaccines based on MSP1 and AMA1 antigens and whole organism sporozoite-based malaria vaccine in Mali. He has been a faculty member for workshops on malaria case management for National Malaria Control Program staff and the health service providers in Mali. He led a five-year malaria incidence study in children in Bandiagara from 2009 to 2014 including geographic information systems-based analyses identifying parasite reservoirs in the community.
Actual work
Currently, Dr Coulibaly is leading: i) a five year incidence study supported by the NIH R01 5R01AI150534, awarded to Dr Coulibaly in 2021-2026, ii) a study of P. vivax infection in Duffy-negative erythrocyte supported by the Laboratory of Malaria and Vector Research (LMVR) National Institute of Allergy and Infectious Diseases (NIAID)/NIH, iii) a Phase 1a clinical trial of malaria vaccine candidate derived from P. falciparum merozoite surface protein 3 associated with CRM manufactured by Vac-4-All.
Selected publications
- Coulibaly D, Guindo B, Niangaly A, Maiga F, Konate S, Kodio A et al., A Decline and Age Shift in Malaria Incidence in Rural Mali following Implementation of Seasonal Malaria Chemoprevention and Indoor Residual Spraying. Am J Trop Med Hyg. 2021 Mar 1: tpmd200622. doi: 10.4269/ajtmh.20-0622.
- Coulibaly D, Travassos MA, Tolo Y, et al. Spatio-Temporal Dynamics of Asymptomatic Malaria: Bridging the Gap Between Annual Malaria Resurgences in a Sahelian Environment. Am J Trop Med Hyg. 2017 Dec;97(6):1761-1769. PMID:29141722
- Noor AM, Kibuchi E, Mitto B, Coulibaly D, Doumbo OK, Snow RW. Sub-National Targeting of Seasonal Malaria Chemoprevention in the Sahelian Countries of the Nouakchott Initiative. PLoS One. 2015 Aug 31;10(8):e0136919. doi: 10.1371/journal.pone.0136919. eCollection 2015. Erratum in: PLoS One. 2015;10(10):e0140414. PubMed PMID: 26322634
- Thera MA, Doumbo OK, Coulibaly D, Plowe CV et al., A field trial to assess a blood-stage malaria vaccine. N Engl J Med. 2011. doi: 10.1056/NEJMoa1008115.
- Agarwal A, Guindo A, Cissoko Y, Taylor JG, Coulibaly D et al., Hemoglobin C associated with protection from severe malaria in the Dogon of Mali, a West African population with a low prevalence of hemoglobin S. Blood. 2000 Oct1;96(7):2358-63. PubMed PMID: 11001883.

Pr Abdoulaye K Kone
Biosketch
Abdoulaye Kassoum Koné, MD, MPH, PhD, is a senior scientist of the Bandiagara Malaria Program (BMP) at the Malaria Research and Training (MRTC) of the University of Sciences, Techniques and Technologies of Bamako, in Mali. He is Associate Professor of Parasitology-Mycology at the Faculty of Medicine and Odonto-Stomatology of Bamako. He has an expertise in severe malaria diagnosis, management and recognizing the ophthalmologic manifestations such as retinopathy in Malian children with cerebral malaria. Dr Kone serves as clinical investigator, clinical coordinator, and principal investigator in the NIH-NIAID sponsored Bandiagara Malaria Project. He draws from a decade of experience in clinical malaria studies including epidemiological and clinical trials. Since 2010 Dr Kone works on cutaneous leishmaniasis epidemiology focused on diagnosis, clinical description, Leishmania species and vectors identification. As Associate Professor at the FMOS, Dr Kone contribute to train undergraduate MDs, and PharmDs, MSc and PhD candidates in Mali.
Dr Kone areas of expertise include malaria epidemiology, clinical trials, Leishmaniasis epidemiology, parasitic diseases and vectors.
Actual work
Is centered on three topics. First topic is leishmaniasis epidemiology. It focuses on clinical presentation, hosts, Leishmania species and Phlebotomus species interactions in Mali. This was done through an investigation of outbreak of cutaneous leishmaniasis in dogon country. This project was supported by the ministry of health in Mali. The second topic of work is on the clinical development of vaccines against malaria and viral infections. The malaria vaccine candidate, the MSP3-CRM candidate manufactured by Vac-4-All is being tested in a series of clinical trials to demonstrate the proof of concept of its safety, immunogenicity and efficacy against clinical malaria. The third topic is on hospital-based epidemiological studies of fungal diseases such as Aspergillosis and Histoplasmosis.
Selected publications
- Kone AK, Niaré DS, Piarroux M, Izri A, Marty P, Laurens MB, Piarroux R, Thera MA, Doumbo OK. Visceral Leishmaniasis in West Africa: Clinical Characteristics, Vectors, and Reservoirs J Parasitol Res. 2019 Sep 2; 2019:9282690. doi: 10.1155/2019/9282690. eCollection 2019. Review. PMID: 31565426
- Kodio A, Coulibaly D, Koné AK, Konaté S, Doumbo S, Guindo A, Bittar F, Gouriet F, Raoult D, Thera MA, Ranque S. Blastocystis Colonization Is Associated with Increased Diversity and Altered Gut Bacterial Communities in Healthy Malian Children. Microorganisms. 2019 Dec 4;7(12). pii: E649. doi: 10.3390/microorganisms7120649. PMID: 31817168
- Kané B, Diallo KW, Koné AK, Simaga T, Koné O, Traoré A, Dicko FT. Paludisme grave chez un nouveau-né prématuré à l’unité de néonatologie de l’Hôpital du Mali. Rev Mali Infect Microbiol 2020, Tome 15 : 69-71
- Ouattara A, Niangaly A, Adams M, Coulibaly D, Kone AK, Traore K, Laurens MB, Tolo Y, Kouriba B, Diallo DA, Doumbo OK, Plowe CV, Djimdé A, Thera MA, Laufer MK, Takala-Harrison S, Silva JC. Epitope-based sieve analysis of Plasmodium falciparum sequences from a FMP2.1/AS02A vaccine trial is consistent with differential vaccine efficacy against immunologically relevant AMA1 variants. Vaccine. 2020 Jul 31;38(35):5700-5706. doi: 10.1016/j.vaccine.2020.06.035. Epub 2020 Jun 20.
- Bogale HN, Cannon MV, Keita K, Camara D, Barry Y, Keita M, Coulibaly D, Kone AK, Doumbo OK, Thera MA, Plowe CV, Travassos M, Irish S, Serre D. Relative contributions of various endogenous and exogenous factors to the mosquito microbiota. Parasit Vectors. 2020 Dec 10;13(1):619. doi: 10.1186/s13071-020-04491-7.
- Nabet C, Kone AK, Dia AK, Sylla M, Gautier M, Yattara M, Thera MA, Faye O, Braack L, Manguin S, Beavogui AH, Doumbo O, Gay F, Piarroux R. New assessment of Anopheles vector species identification using MALDI-TOF MS. Malar J. 2021 Jan 9;20(1):33. doi: 10.1186/s12936-020-03557-2.

Dr Karim Traore
Biosketch
Dr Karim Traore is a medical doctor with a PhD degree in biology, he is also a specialist in infectious diseases and anthropology. Dr Traore is a senior scientist of Bandiagara Malaria project at Malaria research and training center, faculty of medicine and dentistry of Mali. Since 2003, Dr Traore is involved in malaria clinical trials for vaccine and drug development, and epidemiological studies in both adult and children. The area of expertise of Dr Traore include epigenetics, interaction of environment and human genetic in susceptibility to infectious diseases. As senior researcher, Dr Traore is also involved in teaching parasitology and mycology, and ethics at the university of Sciences, Techniques, and Technologies of Bamako, and the training of MDs and master’s degree students. Dr Traore has been the coordinator of an ethic consortium involving Mali, Ghana, Guinea, and the catholic university of Lyon in France funded by EDCTP. Une image contenant personne, complet, homme, souriant Description générée automatiquement
Dr Traore earned his MD degree at the school of medicine and pharmacy of Mali in 2002, then successively completed a master’s degree in anthropo-biology, a diploma of specialization (DES) in infectious diseases at the university Cheick Anta Diop of Dakar in Senegal in 2011 and 2012, and a PhD degree at the university of Lyon, France in 2018. Since 2020 Dr Traore is an EDCTP senior fellow.
Actual work
The current work of Dr Traore include:
Senior fellowship of EDCTP funded program on research capacity development (TMA2018SF-2471), 2020-24. Under this senior fellowship, Dr Traore is conducting a clinical trial to evaluate the safety and efficacy of dihydroartemisinine plus piperaquine compared to sulfadoxine pyrimethamine in malaria chemoprevention in school-aged children in Bandiagara. Senior fellowship also includes the training and mentoring of MD candidate, master and PhD students.
Clinical coordinator of a phase 1a malaria vaccine trial testing a vaccine candidate derived from P. falciparum merozoite surface protein 3 associated with CRM manufactured by Vac-4-All from 2021 to present.
Co-investigator of pediatric malaria incidence studies from 2021 to present.
Clinical Monitor of clinical trials and epidemiological studies being conducted or coordinated by MRTC
Teaching parasitology -mycology, and ethics
Selected publications
- Traore K, Konate S, Thera MA, Niangaly A, Ba A, Niare A, Di Cristofaro J, Baby M, Picot S, Chiaroni J, Boetsch G, Doumbo OK. (2019) Genetic polymorphisms with erythrocyte traits in malaria endemic areas of Mali. PLoS ONE.2019; 14(1): e0209966.
- Traore K, Thera MA, Bienvenu A-L, Arama C, Bonnot G, Lavoignat A, Doumbo OK, Picot S. Interaction between environment, nutrient-derived metabolites, and immunity: A possible role in malaria susceptibility/resistance in Fulani and Dogon of Mali. PLOS ONE. 2017;12(12):e0189724.
- Maiga H, Grivoyannis A, Sagara I, Traore K, Traore OB, Tolo Y, Traore A, Bamadio A, Traore ZI, Sanogo K, Doumbo OK, Plowe CV, Djimde AA. Selection of pfcrt K76 and pfmdr1 N86 Coding Alleles after Uncomplicated Malaria Treatment by Artemether-Lumefantrine in Mali. Int J Mol Sci. 2021 Jun 3;22(11):6057. doi: 10.3390/ijms22116057. PMID: 34205228; PMCID: PMC8200001.
- Thomson-Luque R, Votborg-Novél L, Ndovie W, et al. Plasmodium falciparum transcription in different clinical presentations of malaria associates with circulation time of infected erythrocytes. Nat Commun. 2021 Jul 30;12(1):4711. doi: 10.1038/s41467-021-25062-z. PMID: 34330920; PMCID: PMC8324851
- Traore K, Bull* S, Niare A, Konate S, Thera MA, Kwiatkowski D, et al. Understandings of genomic research in developing countries: a qualitative study of the views of MalariaGEN participants in Mali. BMC Med Ethics 2015; 16 (1):42.
- Traore K, Lavoignat A, Bonnot G, Sow F, Bess GC, Chavant M, et al. Drying anti-malarial drugs in vitro tests to outsource SYBR green assays. Malar J. 2015;14(1):90.
Other staff
| Junior Scientists | Biologists / Lab Technicians Data Manager Admin & Financial Manager Drivers |
Field Workers Support Staff |
|---|---|---|
|
Bouréma Guindo, MD, MSc candidate Bourama Tangara, MD, MSc candidate Ali Thera, MD, MSc candidate Aïchatou Diawara, MD, MSc candidate Mohamed Cissé, MD, MSc candidate Astou Diallo, PharmD, MSc candidate Moctar Coulibaly, MD Amidou Traoré, MD Souleymane Traoré, MD Kindié Kouriba, MD |
Biologists / Lab Technicians Fayçal Maïga, BSc Ladji Diarrassouba, BSc Data Manager Ibrahima Kébé, MSc Administrative and Financial Manager Sékouba Mariko, MBA Drivers Abinem Doumbo Moussa Niaré |
Field Workers Boucari Djiguiba Abdoulaye Ouologuem Paul Dougnon Harouna Doumbia Rokia Traoré Halima Coulibaly Support Staff Issa Tapily Sadio Dolo Saidou Karembé Domo Ouologuem Ampouroli Dougnon Soliba Koroma |
Organizational chart